COVID-19: THE MICROARRAY MATTERS
Rapidly detect SARS-CoV-2 and its evolving variants.

PathogenDx Microarray technology delivers industry-leading sensitivity and specificity for SARS-CoV-2 (COVID-19) virus testing. Our technology is supported by the Rapid Acceleration of Diagnostics (RADx) Initiative, from the National Institutes of Health.
“The EnviroX-RV rapidly detected SARS-CoV-2 RNA on environmental swabs from stainless steel surfaces, outperforming the modified CDC 2019-Novel Coronavirus Real-Time RT PCR diagnostic panel.”
– Scott Coates, Senior Director, AOAC Research Institute
Multiplexed Testing Advantages.
Capability: Multiplexed RT-PCR + DNA microarray – DetectX-Rv: For identification of SARS-CoV-2 (N1, N2) and RNase P gene.
Specificity: Sensitive and
specific, qualitative detection
of SARS-CoV-2.
specific, qualitative detection
of SARS-CoV-2.



Throughput: The unique nature of our multiplexed array allows for testing for 96 tests per kit, with additional throughput up to 384 samples using continuous processing and automation.
Ultrarapid: Results in 6-8 hours from
receipt of the sample.
receipt of the sample.
Applications: Works with all approved nasopharyngeal swabs, nasal
aspirate/fluid, and viral DNA/RNA purification kits.
aspirate/fluid, and viral DNA/RNA purification kits.

MULTIPLEXED VIRAL DIAGNOSTIC ASSAY FOR THE DETECTION OF SARS-CoV2.
The global pandemic has impacted the entire world. With class-leading DNA-based microarray testing technology to our credit, PathogenDx has rapidly developed Detectx-Rv to contribute to viral detection.
This advanced diagnostic solution utilizes nasopharyngeal swabs and nasal aspirate/fluid, and viral RNA purification kits, combining RT-PCR with DNA microarray for sensitive and specific, qualitative detection. It is effective in testing on samples collected from individuals with earliest signs and symptoms of infection who are suspected of COVID-19.
Detectx-Rv contains up to 12 specific probes—more than that of any current FDA authorized COVID-19 test, and will differentiate Corona-like-viruses from SARS-CoV-2 delivering industry leading accuracy.
- This product has not been FDA cleared or approved, but has been authorized for emergency use by FDA under an EUA for use by authorized laboratories.
- This product has been authorized only for the detection of nucleic acid from SARS-CoV-2, not for any other viruses or pathogens.
- The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 360bbb- 3(b)(1), unless the declaration is terminated, or authorization is revoked sooner.

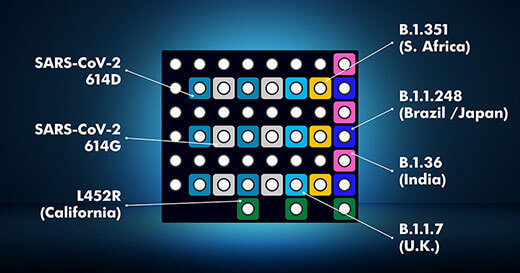
MULTIPLEXED DIAGNOSTIC ASSAY FOR RAPID DETECTION OF SARS-CoV-2 CLADE VARIANTS.
Rapid, affordable Variant Detection for SARS-CoV-2 is now available thanks to the PathogenDx Microarray. Our NIH-supported technology provides invaluable insight to guide scientific discovery and public health decision-making—today not someday. And without the time and expense of sequencing.
- All major variants detected
- Variant results in 4 hours after positive sample from a Q-PCR test
- Run up to 576 samples in a single shift
- A fraction of the cost of NGS Sequencing
COVID-19 ENVIRONMENTAL DETECTION
Envirox-Rv is an air and surface swab test to ensure spaces are virus free.

AEROSOLIZED AND SURFACE SWAB ENVIRONMENTAL MONITORING TEST.
SARS-CoV-2 (COVID-19) can persist on surfaces and in the air, putting humans at risk of transmission. COVID-19 viral RNA is known to be present in air for 3 hours and up to 2-3 days on surfaces. Environmental services personnel are a limited resource to combat this. Rapid identification of contaminated air and surfaces enable targeted environmental interventions and prioritization of environmental services to the highest risk locations.
The Envirox-Rv test combines the power of our Detectx-Rv platform with best-in-class bioaerosol and surface swab collection technology to detect viral contamination. Envirox-Rv is based on RT-PCR + DNA microarray hybridization for qualitative detection of the capture of nucleic acid from the 2019-nCoV (COVID-19)— as well as a host of other viral organisms present in the air and surfaces of the surrounding environment.
The unique aspect of the Envirox-Rv test is high collection efficiency and analysis of aerosolized viral and fungal contamination, with a collection capability in a liquid output that can be rapidly analyzed within an 8-hour shift. The Envirox-Rv test provides superior sensitivity and unique analyte specificity compared to conventional RT-PCR by targeting three (3) SARS-CoV-2 loci, that includes equivalent mis-matching probes to ensure accuracy and certainty in the result.
ENVIROx–Rv TEST KIT: Microarray Slides for 96 Swab Samples; Reagents; Positive and Negative Controls.
WHY YOU SHOULD BE TESTING AEROSOLIZED AND SURFACE SWABS FOR COVID-19:
This test is for Research Use Only. It is NOT for Diagnostic Use.