SYNDROMIC LEVEL MULTIPLE TARGETS IN A SINGLE-WELL ARRAY TEST.
New assays powered by the PathogenDx D3 Array™ will deliver a more flexible, efficient and lower cost path to detection in clinical health
UTI TESTING WITH A DIFFERENCE
PathogenDx is harnessing the power of its D3 Array™ to provide RUO Assays that deliver more actionable information in a single experiment. Discover how this new technology aids in the understanding of UTIs and appropriate therapies.

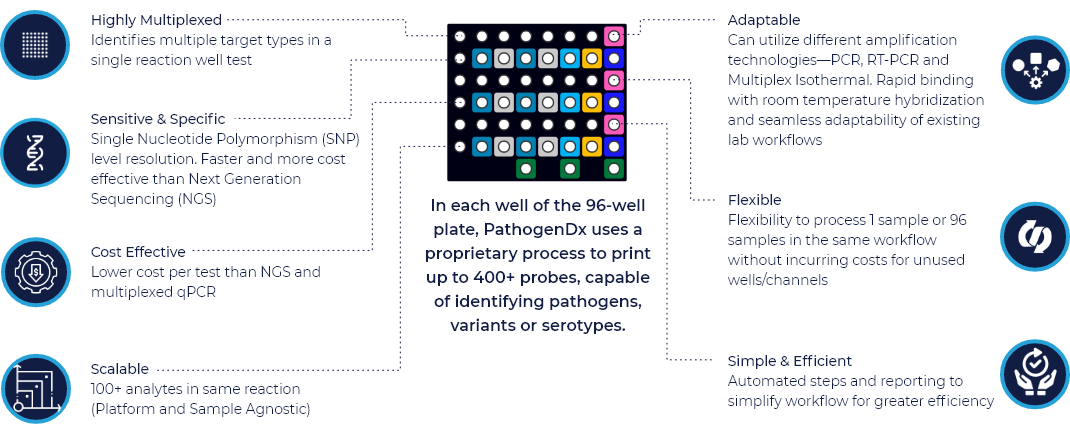
In each well of the 96-well plate, PathogenDx uses a proprietary process to print greater than 300 probes, capable of identifying different pathogens, variants or serotypes—with novel binding of DNA when pathogens are present.


now available!

UTI DETECTION & ANTIBIOTIC RESISTANCE POWERED BY NOVEL MULTIPLEXED MOLECULAR DIAGNOSTICS TECHNOLOGY
D3 Array™-UTI is a fast, sensitive multiplexed PCR nucleic acid test that detects 26 pathogens and 12 antibiotic resistance markers—testing in triplicate, with qualitative and quantitative results in the same reaction AND automated, cloud-based analysis. Support the delivery of rapid UTI treatment with truly disruptive diagnostics.
KEY FEATURES
Gain Ease – Test for 26 causative pathogens +12 Antibiotic Resistance Genes in a single test, with qualitative detection and quantification in the same sample reaction.
Research Efficiently – Utilize only the wells you need for each experiment without incurring cost on the rest of the plate—optimizing reagents and consumables spend.
Streamline Analysis – Reduce the time and cost of deciphering reams of raw data via PDx’s Augury automated, cloud-based data analysis and management.
ASSAYS FOR COVID-19 (EUA)


During the pandemic, PathogenDx rapidly developed DetectX-Rv to contribute to viral detection. Differentiating Corona-like-viruses from SARS-CoV-2 and delivering industry leading accuracy, DetectX-Rv was authorized for emergency use by the FDA under an EUA for use by authorized laboratories.
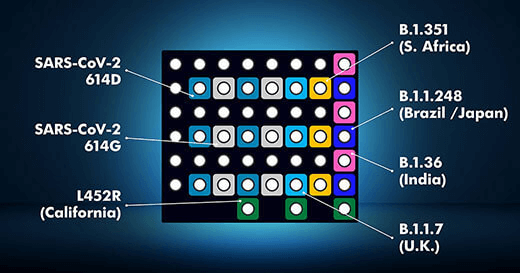
And with the D3 Array™ unique spatial architecture, came the opportunity to develop multiplexed assays for the simultaneous detection of SARS-CoV-2 variants. DetectX-Cv detects all major variants with results in 4 hours after a positive sample.
OTHER D3 ARRAY™ RUO ASSAYS IN THE WORKS.


PathogenDx D3 Array™ unique spatial architecture enables comprehensive, multiplexed assays for the study of and simultaneous detection of a wide range of pathogens associated with Women’s Health including the detection of HPV variants, vaginal infections, and STIs. The D3 Array™ format, with enhanced probe binding, single gene copy sensitivity, competitive economies and rapid time-to-result, is well suited for this need.
The D3 Array™ promotes confidence in your results. The simultaneous detection of a wide range of pathogens aids in rapid analysis of specimens. PathogenDx is committed to supporting women’s health initiatives by providing accurate and sensitive tests. Learn more »
Rapid, multiplexed detection of the causative agent(s) of bloodborne infections and the bacteria’s antimicrobial susceptibility are critical for the selection of treatment options. With traditional ID/AST testing, time to result can be days and costly antibiotics are often prescribed until the specific pathogen can be identified and treatment optimized.
PathogenDx’s D3 Array™ has the potential to provide highly-multiplexed detection and antibiotic resistance information in a low-cost, rapid format that has the potential to significantly increase quality of care by providing critical information to researchers and physicians, with the promise of reducing morbidity and mortality in bacteremic patients. Watch for such future products. Learn more »